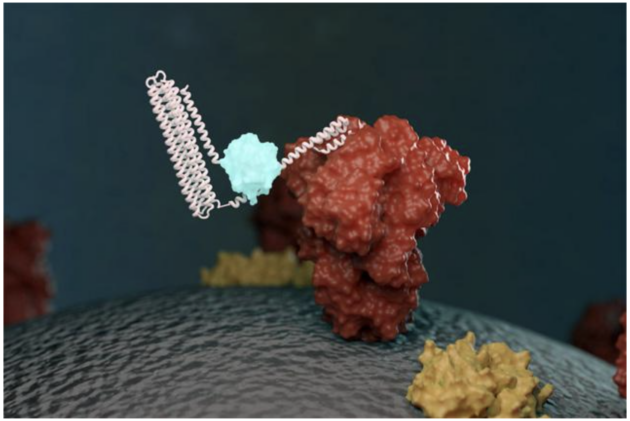
 Illustration of a biosensor glowing (light blue) after detecting its target (a coronavirus protein, red). (UW IPD | Ian Haydon)
Illustration of a biosensor glowing (light blue) after detecting its target (a coronavirus protein, red). (UW IPD | Ian Haydon)
A new biotech startup has hatched from the University of Washington’s Institute for Protein Design with $6 million in funding and a leadership team fresh from Neoleukin Therapeutics, another IPD spinout.
Monod Bio is developing biosensors that can quickly detect viral proteins, toxins, antibodies, or other molecules. The sensors are built from engineered proteins developed at the IPD. When the sensor recognizes its target, it emits light.
The sensors are engineered using IPD software that can design proteins from scratch, “de novo”, instead of from the more limited number of designs found in nature.
“We are the first company, as far as we know, using de novo computational protein design for biosensors and diagnostics,” said chief scientific officer Alfredo Quijano Rubio in an interview with GeekWire. The sensors work in test tubes, and one of the company’s next steps is to make them easier to use, similar to at-home pregnancy tests.
Quijano Rubio designed sensors to recognize a variety of targets while working in the lab of IPD head David Baker, where he was a graduate student until this spring.
In a study in Nature this January, Quijano Rubio, Baker and their colleagues showcased sensors that recognize the “spike” protein of the COVID-19 virus as well as antibodies to it. Other sensors recognized botulism toxin, the cancer-related HER2 receptor, and cardiac troponin, which is released by the body after a heart attack.
“The consequence of this is that you can now bring biosensors to places where you usually don’t have them, like wearables or point of care,” said Monod CEO Daniel-Adriano Silva.
Troponin, for instance, is usually measured in the hospital to help assess if someone has had a heart attack. Instead, Silva envisions testing someone in the ambulance on the way to the hospital.
 Monod CEO Daniel-Adriano Silva, CSO Alfredo Quijano Rubio, and COO David Shoulz. (Monod Photos)
Monod CEO Daniel-Adriano Silva, CSO Alfredo Quijano Rubio, and COO David Shoulz. (Monod Photos)
Silva left his job this spring as head of research at Neoleukin, which is developing treatments for cancer and COVID-19, to explore starting a new company. “I started going back to talk to David, to see what was the next technology,” he said.
He didn’t have to look far. Quijano Rubio is also a co-founder at Neoleukin. David Shoultz, formerly head of Neoleukin’s COVID-19 program, was tapped as COO and co-founder.
Other Monod co-founders include IPD head Baker, IPD scientist Andy (Hsien Wei) Yeh and Dave Johnson, former CEO of VelosBio, which was acquired by Merck for $2.75 billion last year.
The team has been operating since November from CoMotion Labs on the University of Washington campus. In December, the startup completed its $6 million seed round from friends and family.
Seattle is peppered with IPD startups. Cyrus Biotechnology, for instance, recently raised $18 million and has more than 90 drug company partnerships, A-Alpha Bio has raised $20 million, and Icosavax went public this year with a $180 million raise to advance its vaccines for COVID-19 and other conditions.
RELATED: Univ. of Washington AI protein folding discovery wins ‘Breakthrough of the Year’ award from Science
IPD researchers also won Science magazine’s “Breakthrough of the Year” award this December for their AI-powered software to predict how proteins fold. Monod’s sensors are designed with protein engineering software developed earlier at IPD, called Rosetta.
The Monod team will use the funds to mature and expand its platform, generate new biosensors for additional targets in biotechnology and health, and build prototypes to detect targets in real-world applications.
IPD researchers have also built sensors to detect a suite of COVID-19 antibodies, with the aim of determining a person’s level of antibody immune protection. They showcased these sensors in a recent preprint.
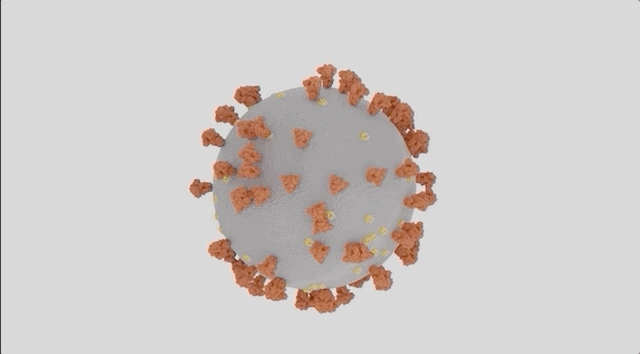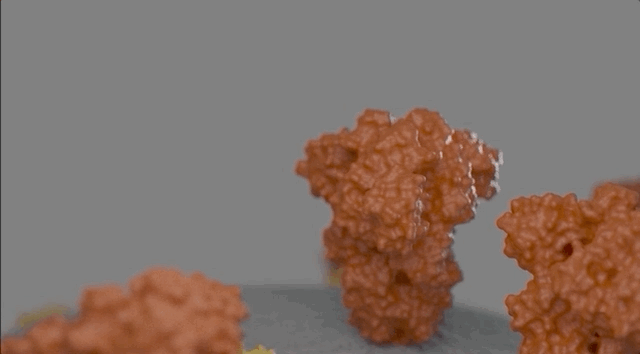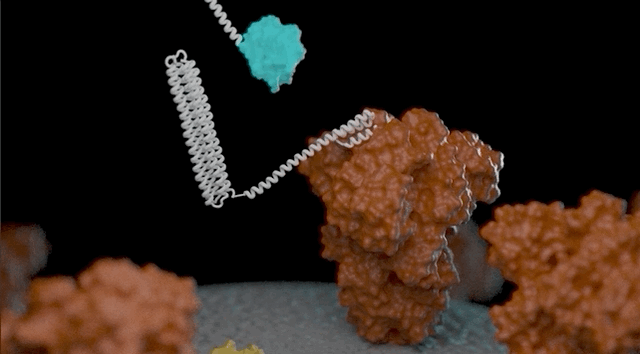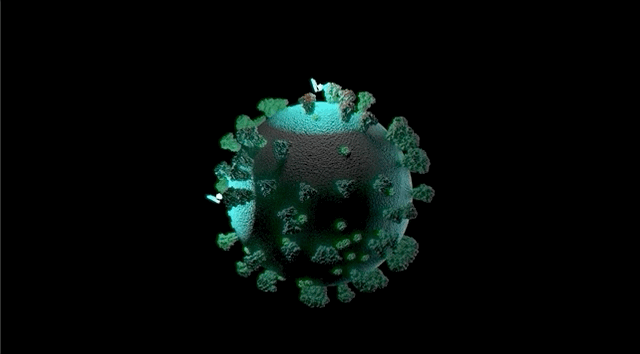 Video of a biosensor binding its target (a coronavirus protein, red) and then glowing. (UW IPD Video)
Video of a biosensor binding its target (a coronavirus protein, red) and then glowing. (UW IPD Video)
Monod’s sensors rely on two components. One component is a protein that recognizes a target. Upon target recognition the protein shifts from a closed to an open state. In its open state, the protein is able to bind the second component of the sensor. When the two bind, the sensor lights up. The light is emitted from luciferase, a light-emitting enzyme.
The region that recognizes the target can be swapped out for another. “Alfredo figured out a way to make this in a modular way,” said Silva.
Upon target binding, the sensor shifts in a way that propagates through the protein to open up its shape. Such an effect, where an action at one part of a protein is transmitted to another, is called allostery, and it’s used to regulate the activity of many of the body’s proteins. Allostery was discovered in the 1960’s by Nobel-prize winning biochemist Jacques Monod and his colleagues.
Monod is now hiring protein biochemists and engineers. The team is looking for its own space in Seattle’s tight lab market and aims to grow to around 15 or 20 employees by the end of 2022.






